Klicken Sie auf Lektionenliste an Chemistry - الكيمياء لغات (في الترمين)
Klicken Sie auf dem Seite des Faches an. Chemistry - الكيمياء لغات (في الترمين)
الصف الثاني الثانوي und wählen Sie andere Lektion aus.
Melden Sie sich bei Nafahm und Verfolgen deine Lektionen regelmäßig.

Haben Sie eine Frage zu diesem Thema?
Fügen Sie Ihre Frage hinzu und sehen Sie die hinzugefügten Fragen in dieser Lektion.
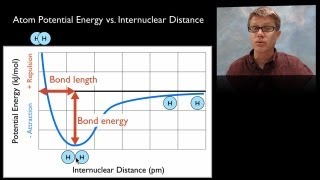
The Covalent Bond It's the formed by sharing electrons between the atoms or the moulecole of the compound there is three main types of covalent which is 1_ pure covalent bond : its the bond formed between two similer nonmetal atoms have the same electronigativity then by subtract there electonegativity the result will became zero 2_ non-polar covalent bond : its the bond formed between two diffrente nonmetals and they have a diffrente electronegativity so by subtract there elecronegtivty iy will give us 0.4 or less 3_polar covalent bond its the bond formed between two diffrinte non metal atom to suctract there electronecativity will give 1.7 or less
:Die Zusammenfassung bearbeitet von Mona Ibrahim



























Kommentare